Next-Generation Targeted Protein Degradation: Beyond Classical PROTACs
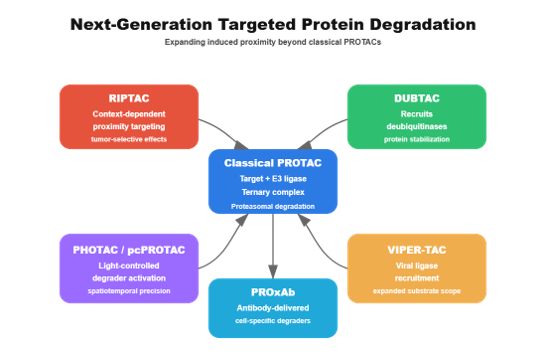
Figure: Evolution of induced proximity therapeutics beyond classical PROTACs. Classical PROTACs recruit an E3 ubiquitin ligase to a target protein to induce degradation. Next-generation modalities expand this principle: RIPTACs create context-dependent proximity for tumor-selective lethality; DUBTACs stabilize proteins by recruiting deubiquitinases; PHOTACs and pcPROTACs enable light-controlled degradation; VIPER-TACs leverage viral ligases to broaden substrate scope; and PROxAb systems use antibody delivery to achieve cell-type-specific degrader activation.
Executive Overview
Targeted protein degradation (TPD) has reshaped modern drug discovery by shifting the paradigm from occupancy-driven pharmacology to event-driven pharmacology. Rather than inhibiting protein function through sustained binding, degraders trigger the removal of disease-driving proteins from the cell entirely. This catalytic mode of action enables suppression of scaffolding proteins, transcription factors, and other historically “undruggable” targets.
First-generation PROTACs (proteolysis-targeting chimeras) demonstrated that small molecules can recruit E3 ubiquitin ligases to a target protein, inducing ubiquitination and proteasomal degradation. Clinical progress in oncology validated the approach. However, the field has rapidly evolved beyond classical PROTAC architectures.
Today’s next-generation modalities expand induced proximity in multiple dimensions:
- Context-dependent proximity (RIPTACs)
- Targeted protein stabilization (DUBTACs)
- Light-controlled degraders (PHOTACs, pcPROTACs)
- Virus-inspired proximity systems (VIPER-TACs)
- Antibody-delivered degraders (PROxAb)
Collectively, these approaches redefine TPD as a broader class of induced proximity therapeutics. The future is no longer just about degradation — it is about programmable control of protein fate, function, and location.
The Induced Proximity Paradigm
From Inhibition to Event-Driven Pharmacology
Traditional small molecules rely on occupancy: a drug must remain bound to exert effect. In contrast, degraders operate catalytically. Once a target is ubiquitinated and committed to degradation, the degrader molecule can, in principle, dissociate and engage another copy of the protein.
This creates several advantages:
- Reduced dependence on high-affinity binding
- Ability to eliminate scaffolding functions
- Suppression of both enzymatic and non-enzymatic activity
- Potential to overcome resistance mutations
At the core of TPD is induced proximity — the deliberate forced interaction between two proteins that would not normally engage. In classical PROTACs, the two components are:
- The target protein
- An E3 ubiquitin ligase
A heterobifunctional molecule brings them together, forming a ternary complex that drives ubiquitination.
However, induced proximity is not limited to E3 ligases or degradation. The same logic can recruit:
- Deubiquitinases
- Kinases
- Autophagy initiators
- RNases
- Viral ligases
- Conditional control elements
This conceptual shift is what defines the next wave of innovation.
RIPTACs: Context-Dependent Induced Proximity
Mechanistic Principle
RIPTACs bind:
- A broadly expressed essential protein
- A tumor-restricted or overexpressed protein
By forcing proximity, the essential protein becomes functionally inactivated — but only in cells expressing the tumor-specific partner. This produces synthetic-lethal-like selectivity without directly targeting mutated genes.
Why This Matters
Many essential proteins are poor therapeutic targets due to systemic toxicity. RIPTACs introduce a spatial and contextual filter: only cells co-expressing both proteins are affected.
This approach offers:
- Precision oncology selectivity
- Potential activity against non-mutated drivers
- Reduced reliance on enzymatic inhibition
Challenges
- Identifying suitable tumor-restricted anchors
- Avoiding off-tumor expression
- Managing adaptive rewiring
RIPTACs illustrate how induced proximity can move beyond degradation into conditional functional rewiring.
DUBTACs: Targeted Protein Stabilization
While most proximity-based drugs remove proteins, DUBTACs (Deubiquitinase-Targeting Chimeras) reverse this logic.
Mechanism
DUBTACs recruit a deubiquitinase (DUB) to a ubiquitinated target protein. This removes ubiquitin chains and rescues the protein from degradation.
Why Stabilization Matters
Many diseases arise from insufficient protein levels:
- Tumor suppressor loss
- Haploinsufficiency disorders
- Misfolding-related degradation
Instead of inhibiting E3 ligases globally (which risks toxicity), DUBTACs localize deubiquitination to a specific protein.
Applications
- Stabilizing mutant but functional tumor suppressors
- Correcting degradation-prone variants
- Potential rare disease interventions
Key Challenges
- DUB selectivity and off-target deubiquitination
- Achieving productive ternary complexes
- Avoiding interference with endogenous ubiquitin signaling
DUBTACs demonstrate that induced proximity is not inherently degradative — it can restore function.
Conditional and Controllable Degraders
Precision control is an emerging frontier. Light-responsive degraders introduce temporal and spatial specificity.
PHOTACs: Photoswitchable PROTACs
PHOTACs incorporate photoswitchable moieties (e.g., azobenzene-like systems) that toggle between active and inactive conformations upon light exposure.
This allows:
- Reversible activation
- Localized degradation
- Dynamic pathway interrogation
Potential applications include neuroscience and developmental biology, where temporal control is critical.
pcPROTACs: Photocaged PROTACs
Photocaged PROTACs remain inactive until light exposure removes a blocking group. Unlike PHOTACs, activation is typically irreversible.
Advantages
- Clean on/off state
- Reduced systemic activity
- High spatiotemporal precision
PHICS and PHORCS
These systems extend light control beyond degradation into proximity-driven signaling or reversible protein complex formation.
Rather than simply removing proteins, they allow:
- Light-triggered protein assembly
- Signal rewiring
- Controlled functional modulation
Collectively, these technologies move TPD toward precision pharmacology.
VIPER-TACs: Viral-Inspired Proximity Systems
Why Viral Ligases?
- Distinct substrate preferences
- Unique regulatory interfaces
- Potential to target proteins inaccessible to canonical ligases
Applications
- Antiviral therapies
- Immune modulation
- Novel substrate scope expansion
The challenge lies in specificity and safety — viral machinery must be carefully engineered to avoid unintended degradation.
PROxAb: Antibody-Delivered Degraders
One of the major limitations of small-molecule degraders is systemic exposure and tissue distribution. PROxAb platforms combine:
- Targeting antibodies
- Cleavable linkers
- Intracellular degrader payloads
How It Works
- Antibody binds a cell-surface antigen
- Complex internalizes
- Linker cleaves intracellularly
- Released PROTAC engages intracellular target
This approach mirrors antibody-drug conjugates (ADCs), but instead of cytotoxic payloads, the cargo is a degrader.
Advantages
- Cell-type specificity
- Reduced systemic toxicity
- Expanded therapeutic window
Technical Hurdles
- Endosomal escape efficiency
- Linker stability
- Drug-to-antibody ratio optimization
PROxAb represents convergence between biologics and TPD.
Design Principles Across Next-Gen Modalities
Despite architectural diversity, common design rules apply.
Ternary Complex Cooperativity
Productive proximity requires:
- Favorable protein-protein interactions
- Proper geometry
- Dynamic stability
Cooperative ternary complex formation often predicts degradation efficiency better than binary binding affinity.
Ligase or Enzyme Selection
Choice of recruited partner dictates:
- Tissue specificity
- Resistance pathways
- Safety profile
Emerging strategies expand beyond CRBN and VHL to tissue-restricted ligases.
Linker Architecture
Linkers determine:
- Flexibility vs rigidity
- Orientation of interacting surfaces
- Cellular permeability
Structure-guided design and AI modeling increasingly guide optimization.
Contextual Selectivity
Next-generation systems incorporate:
- Tumor-restricted expression
- Light activation
- Antibody delivery
- Conditional engagement
Selectivity is shifting from binding specificity to biological context specificity.
Clinical and Translational Landscape
Targeted protein degradation has moved from conceptual novelty to clinical reality, particularly in oncology. Next-generation modalities aim to address:
- Solid tumor resistance
- Central nervous system delivery
- Rare genetic disorders
- Immune modulation
Several biotech companies are now expanding pipelines into:
- Tissue-restricted ligase discovery
- Autophagy-based degradation
- RNA-targeted proximity systems
As the field matures, differentiation increasingly depends on:
- Ligase access
- Conditional control
- Safety margins
- Novel substrate space
Resistance and Evolution of the Field
Resistance mechanisms include:
- E3 ligase downregulation
- Target mutation
- Adaptive rewiring
- Proteostasis compensation
Next-generation platforms attempt to preempt resistance through:
- Alternative ligases
- Conditional activation
- Multi-target strategies
Combinatorial proximity drugs may represent a future frontier.
The Future of Induced Proximity Therapeutics
The evolution of targeted protein degradation reveals a broader principle: drug discovery is becoming programmable biology.
Future directions include:
- Multi-functional chimeras that degrade one protein while stabilizing another
- Tissue-restricted ligase atlases
- AI-designed ternary complexes
- Integration with synthetic lethality frameworks
- Expansion beyond proteins to RNA and chromatin complexes
Ultimately, induced proximity is not a single modality — it is a design philosophy.
The next decade will likely see:
- More precise control
- Broader target classes
- Increased integration with biologics
- Spatial and temporal programmability
Targeted protein degradation began as a clever workaround for undruggable proteins. It is now evolving into a foundational technology platform capable of reshaping therapeutic intervention at a systems level.
The era beyond classical PROTACs has begun — and it is defined not by degradation alone, but by the controlled orchestration of molecular proximity.
Related Services
| Service | |
|---|---|
Small molecule drug discovery for even hard-to-drug targets – identify inhibitors, binders and modulators | |
Molecular Glue Direct | |
PPI Inhibitor Direct | |
Integral membrane proteins | |
Specificity Direct – multiplexed screening of target and anti-targets | |
Express – optimized for fast turn – around-time | |
Snap – easy, fast, and affordable |


